39 electrolysis of water diagram
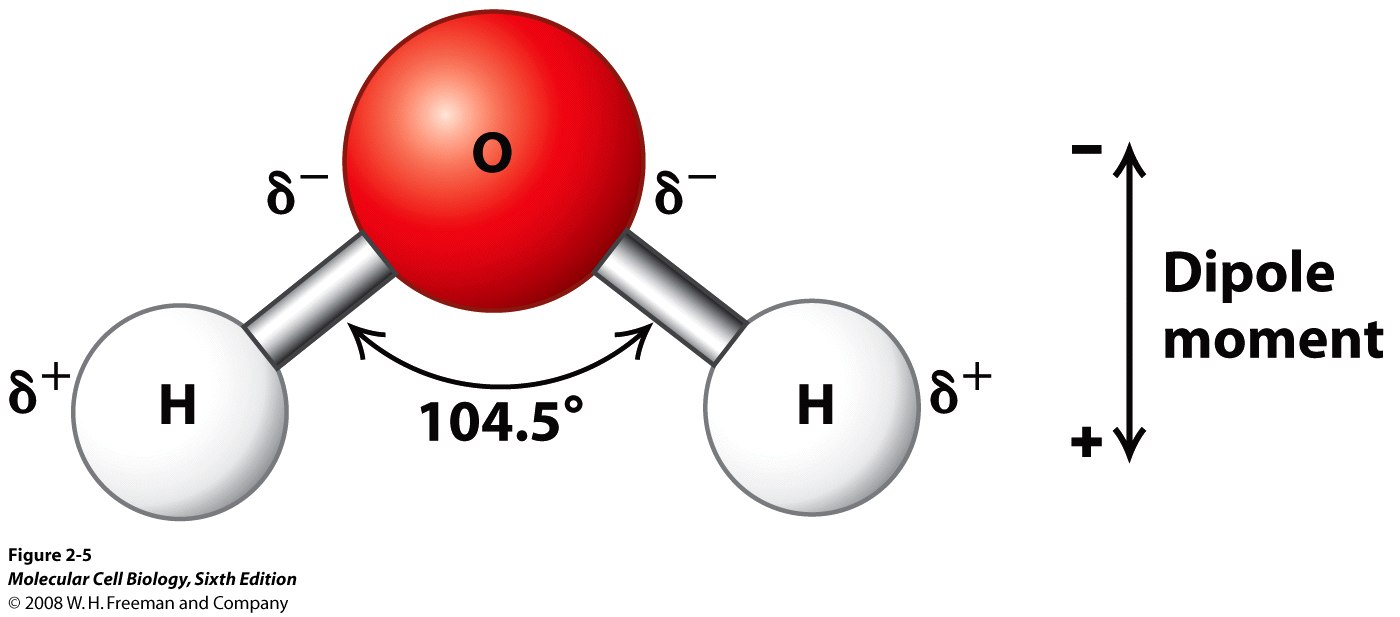
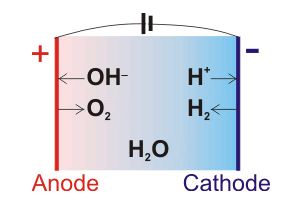
Pourbaix diagram gives the equilibrium regions of water, hydrogen and oxygen at various electrode potentials. Water Electrolysis in the Presence of Salts Salts are 100%dissociate into cations and anions in water and hence increase the ionic concentration for increasing conductivity. When you add salt to the water, the salt ions (which are highly polar) help pull the water molecules apart into ions. Each part of the water molecule (H O) has a charge. The OH- ion is . 2 . negative, and the H+ ion is positive. This solution in water forms an electrolyte, allowing current to flow when a voltage is applied. The H+ ions, called cations, move toward the cathode
30/11/2019 · Hello Everyone.Diagram of Electrolysis of Water || How To Draw Labelled Diagram of Electrolysis of WaterDiagram of Electrolysis of Water, How To Draw Labelle...

Electrolysis of water diagram
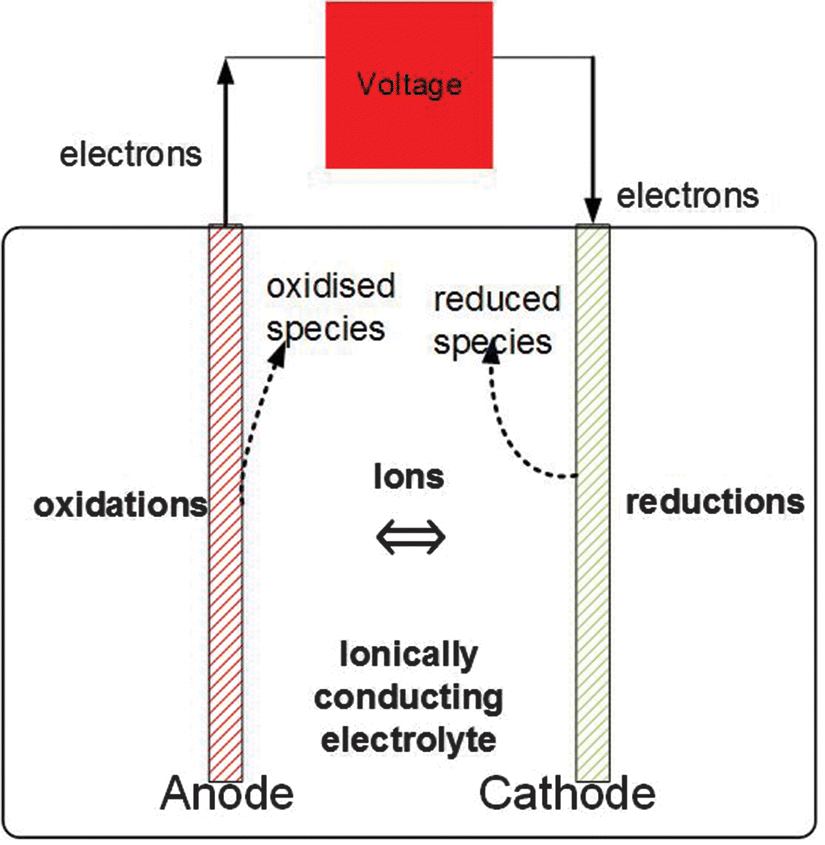
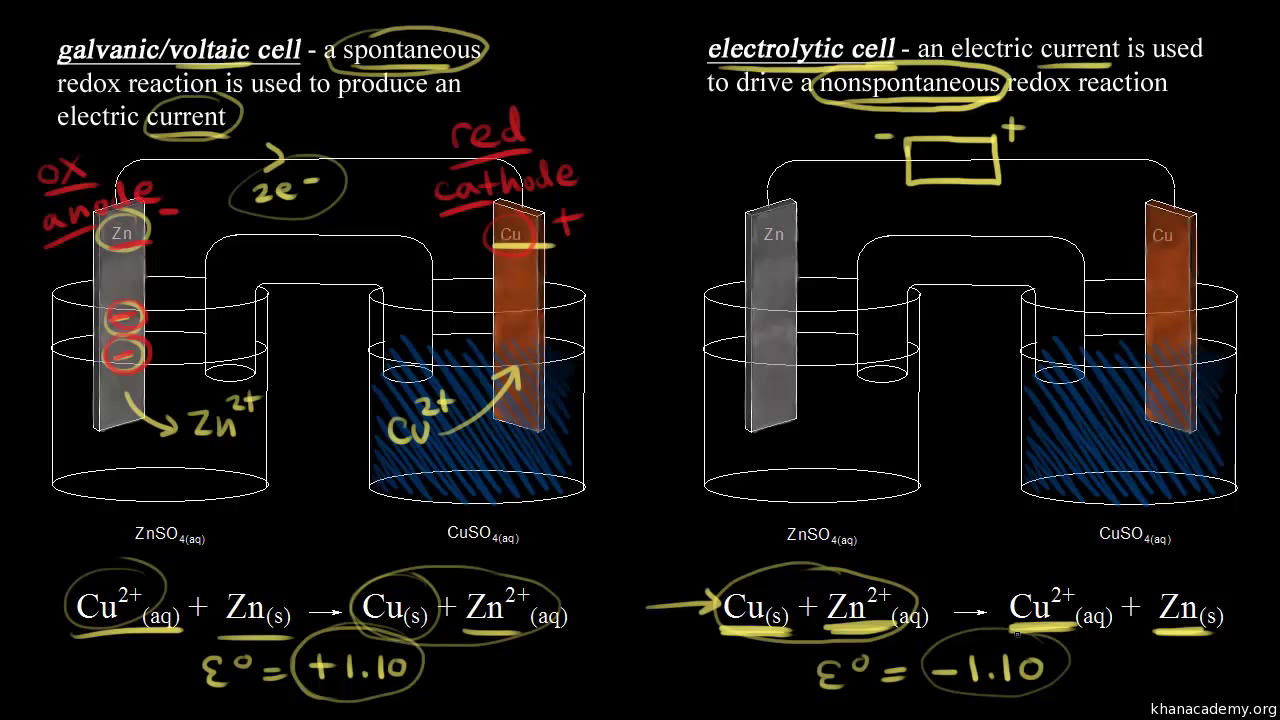
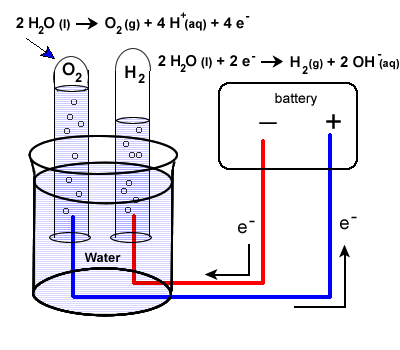
Electric effects on water Magnetic effects on water Electromagnetic effects on water Redox Electrolysis Driving cars using water. Introduction. Passage of an electric current through water causes positive ions, including the inherent hydrogen. Electrolysis of water Electrolysis of water is the decomposition of water (H2O) into oxygen (O2) and hydrogen gas (H2) due to an electric current This electrolytic process is used in some industrial applications when hydrogen is needed. An electrical power source is connected to two electrodes, or... During electrolysis of pure water, at the negatively charged cathode, a reduction reaction takes place, with electrons from the cathode being given to hydrogen cations to form hydrogen gas (the half-reaction balanced with acid): Cathode (reduction): 2H ++2e −→H 2. . …
Electrolysis of water diagram. Electrolysis of water can be achieved in a simple hands-on project, where electricity from a battery is passed through a cup of water (in practice a saltwater Have the students draw a diagram of the experiment set-up (i.e., similar to figure in the "Background" section). On their diagram, indicate which... Step 1: Electrolysis of Water - an Explanation. As everyone knows a water molecule is formed by two elements: two positive Hydrogen ions and one negative Oxygen ion. Let's say the diagram is a literal representation of the mechanism. After a certain amount of time, both the H balloon and the O... The electrolysis of water produces hydrogen and oxygen gases. The electrolytic cell consists of a pair of platinum electrodes immersed in water The electrolyte is necessary because pure water will not carry enough charge due to the lack of ions. At the anode, water is oxidized to oxygen gas and... March 6, 2018 -
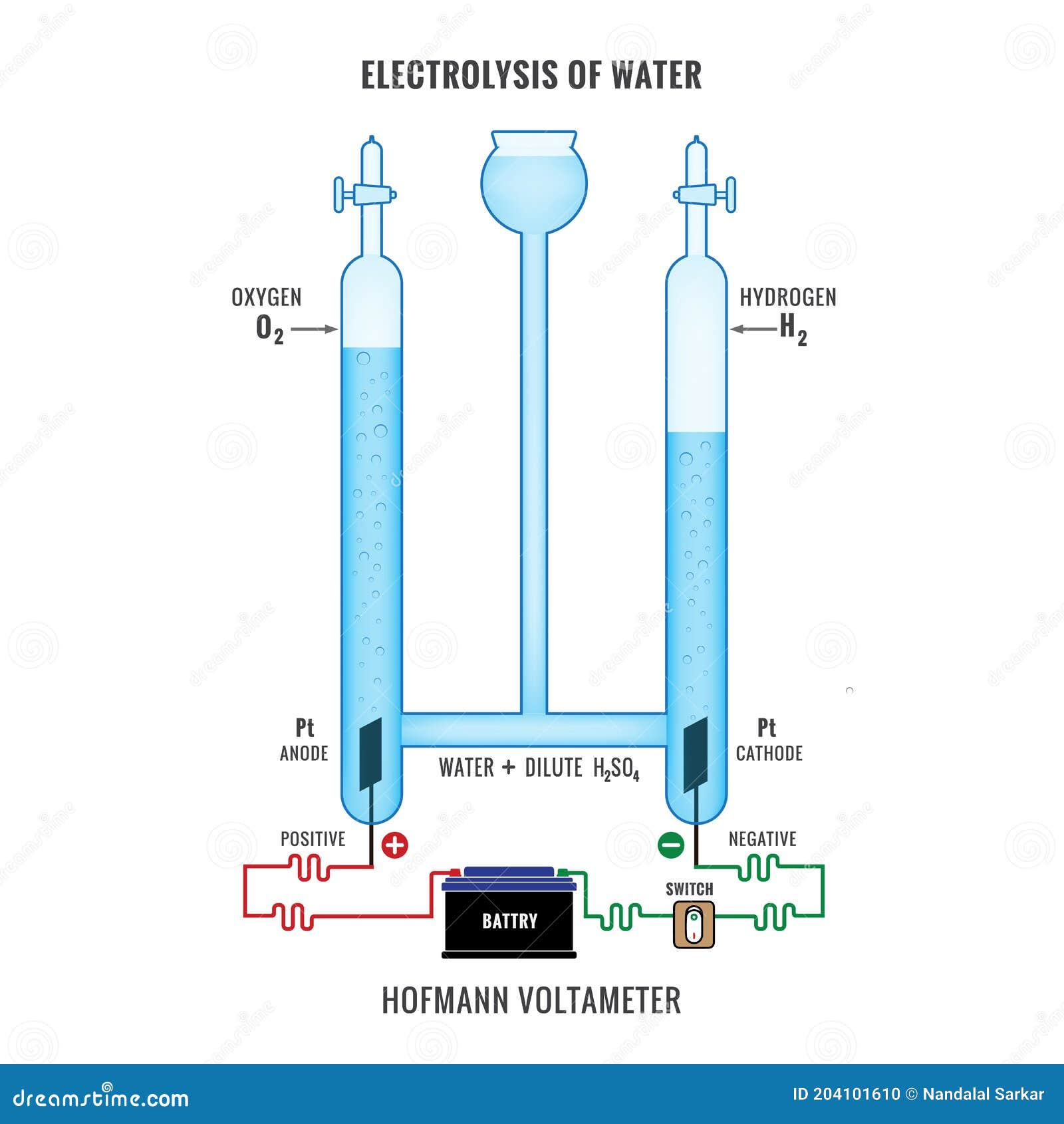
Find Labeled Diagram Show Electrolysis Acidified Water stock images in HD and millions of other royalty-free stock photos, illustrations and vectors in the Shutterstock collection. Thousands of new, high-quality pictures added every day. The diagram shows the electrolysis of molten lithium iodide . 6 (a) (I) Add To Classified 1 Mark . Draw an arrow on the diagram to show the direction of the electron fl ow in the external circuit. answer . correct arrow from negative terminal of battery or from anode; ... water used up; 6 (d) ... July 7, 2021 - Electrolysis is the process of using electricity to split water into hydrogen and oxygen. The reaction takes place in a unit called an electrolyzer. Illustration of Labeled diagram to show the electrolysis of acidified water forming hydrogen and oxygen gases. vector art, clipart and stock vectors. Image 47667616.
28/06/2021 · This video explains How to draw Diagram of Electrolysis of Water in easy steps and compact way. Diagrams In CBSE (NCERT) class 10th are explained in detailed... Pourbaix diagram for water, including equilibrium regions for water, oxygen and hydrogen at STP. The vertical scale is the electrode potential of hydrogen The electrolysis of water in standard conditions requires a theoretical minimum of 237 kJ of electrical energy input to dissociate each mole of water... A simplified diagram of the cell commercially used to produce sodium metal and chlorine gas is shown in Figure 1. Sodium is a strong reducing agent and Figure 2. Water decomposes into oxygen and hydrogen gas during electrolysis. Sulfuric acid was added to increase the concentration of hydrogen... July 13, 2010 - As is the case for other oxidation ... is called a reduction reaction. Using this knowledge, we can add more detail to the previous diagram: The electrolysis of one mole of water produces a mole of hydrogen gas and a half-mole of oxygen gas in their normal diatomic ...
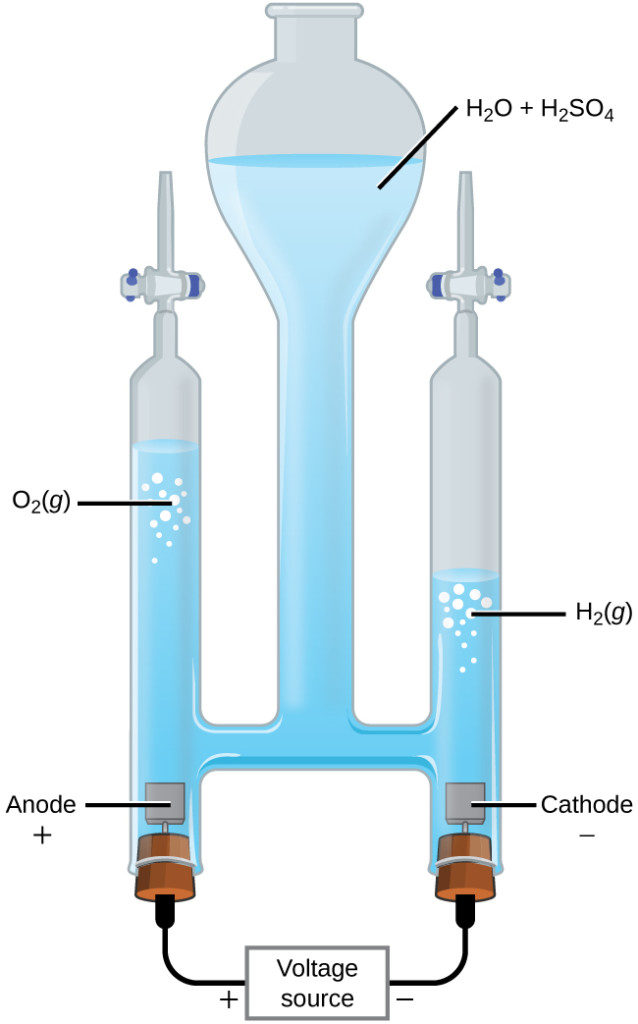
ELECTROLYSIS OF WATER : DIAGRAM - Refer the attached picture.. APPARATUS USED:. Plastic Mug: To carry out the process of electrolysis of water.; Rubber Stopper: To connect Carbon Electrodes with the anode and cathode part of the battery.; Carbon Electrode: To carry out the current discharge in water for electrolysis.; Dilute Sulphuric Acid: For the release of H+ ions easily for electrolysis..
Splitting Water: Electrolysis Experiments + Video. Electrolysis is the process by which an electric current is passed through a substance to Electrolysis is used to remove hair, split compounds, and in the manufacturing process to decorate, strengthen, and make metal surfaces more resistant to rust.
The electrolysis of acidified water. Doc Brown's chemistry revision notes: GCSE chemistry, IGCSE chemistry, O level & ~US grades 9-10 school science courses or equivalent The electrode reactions and products of the electrolysis of acidified water are illustrated by the theory diagram above.
Pourbaix diagram for water, including equilibrium regions for water, oxygen and hydrogen at STP. The vertical scale is the electrode potential of hydrogen or High-pressure electrolysis is the electrolysis of water with a compressed hydrogen output around 12-20 MPa (120-200 Bar, 1740-2900 psi).[14]...
Look at the diagram which shows electrolysis of water and answer the following question What is produced from a positive carbon electrode anode A Oxygen B Carbon dioxide C Ozone D Carbon monoxide
Electrolysis of water is a process in which electricity is used to decompose water into oxygen and hydrogen gas. Diagram of Electrolysis of Water is as shown below. Was this answer helpful? 0 (0) (0) (0) Choose An Option That Best Describes Your Problem. Answer not in Detail.
Electrolysis of water set up. Set up the apparatus as shown in the diagram above. Add a few drops of concentrated sulphuric acid to the water and stir. Allow the current to pass through the water. Adjust the current on the ammeter until there is a gentle evolution of gases at both electrodes.
May 18, 2019 -
Temperature Electrolysis IMECE 2008 J. E. O’Brien ... q ” heat flux, W/cm2 Q ... Consider the process diagram shown in Fig. 2. Water enters the control volume from the left. Since the ultimate feedstock for any large-scale water-splitting operation will be liquid water, it is reasonable to consider the case in
LAB: Electrolysis of Water Electrolysis Apparatus 2H2O 2H2 + O2 Water is a polar molecule meaning that one end (the hydrogen atoms) are positively charged (+) and the other end (oxygen side) is negatively (-) charged. The negative and positive charges on the ends of a battery can be used to...
Electrolysis of water for energy storage is being very actively pursued. Once generated, there are numerous uses for hydrogen in electrical and natural gas grid, mobility, biogas , and fertilizer applications. Due to their higher operating current densities, high-pressure output...
Figure 2. ∆G-T diagram of water splitting. Because of the endothermic reaction the energy that is equal to ΔH should be provided to split water into hydrogen and oxygen; ΔG as a useful work and TSΔ as a thermal GT diagram for the water splitting reaction is shown in Figure 2. The water decomposition reaction has a large positive free ...
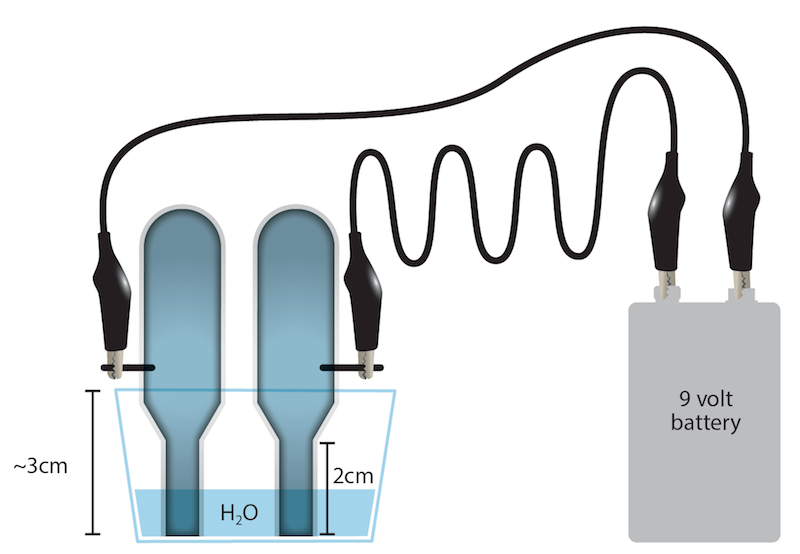
Part 1 – Electrolysis of water In this section, you will use electricity to split apart water molecules! 1. Set up the electrolysis apparatus as shown in the picture. a) Pierce two round holes in the bottom of a plastic cup. (Figure 1a) b) Sharpen two wooden pencils on both ends (after pulling off the eraser). Insert the pencils into the
The diagram below shows the apparatus. ... CuCl2 dissolved in 1 dm3 of water. The students used 50 cm3 of copper chloride solution in each experiment. ... €€€€€A student measured the volumes of each gas produced during the electrolysis of water.
2. Theory of water electrolysis The electrolysis of water is considered a well-known principle to produce oxygen and hydrogen gas. Ultrapure water is fed to the anode structure of the electrolysis cell which is made of porous titanium and activated by a mixed noble metal oxide catalyst.
Electrolysis of water, illustration. Electrolysis is the use of an electrical current to decompose a chemical, in this case water.
Alkaline water electrolysers are designed and manufactured for a wide range of applications. Major gas-consuming industries are: (1) electric power generator cooling in power plants, (2) semiconductor industry, (3) flat panel computers and television screen producing units and (4) glass plants and metallurgical industries.
Electrolysis of Water. This lab will go in your lab book. Follow the directions below to complete the lab. Draw a diagram of your lab set-up (exactly as it appears in this lab). Label the positive and negative end of the battery, show the flow of electrons, label the anode and the cathode, show the...
Alkaline water electrolysis is one of the easiest methods for hydrogen production, offering the advantage of simplicity. The challenges for widespread use of water electrolysis are to reduce energy consumption, cost and maintenance and to increase reliability, durability and safety.
Electrolysis of water is the decomposition of water into oxygen and hydrogen gas due to the passage of an electric current. This technique can be used Pourbaix diagram for water, including equilibrium regions for water, oxygen and hydrogen at STP. The vertical scale is the electrode potential of a...
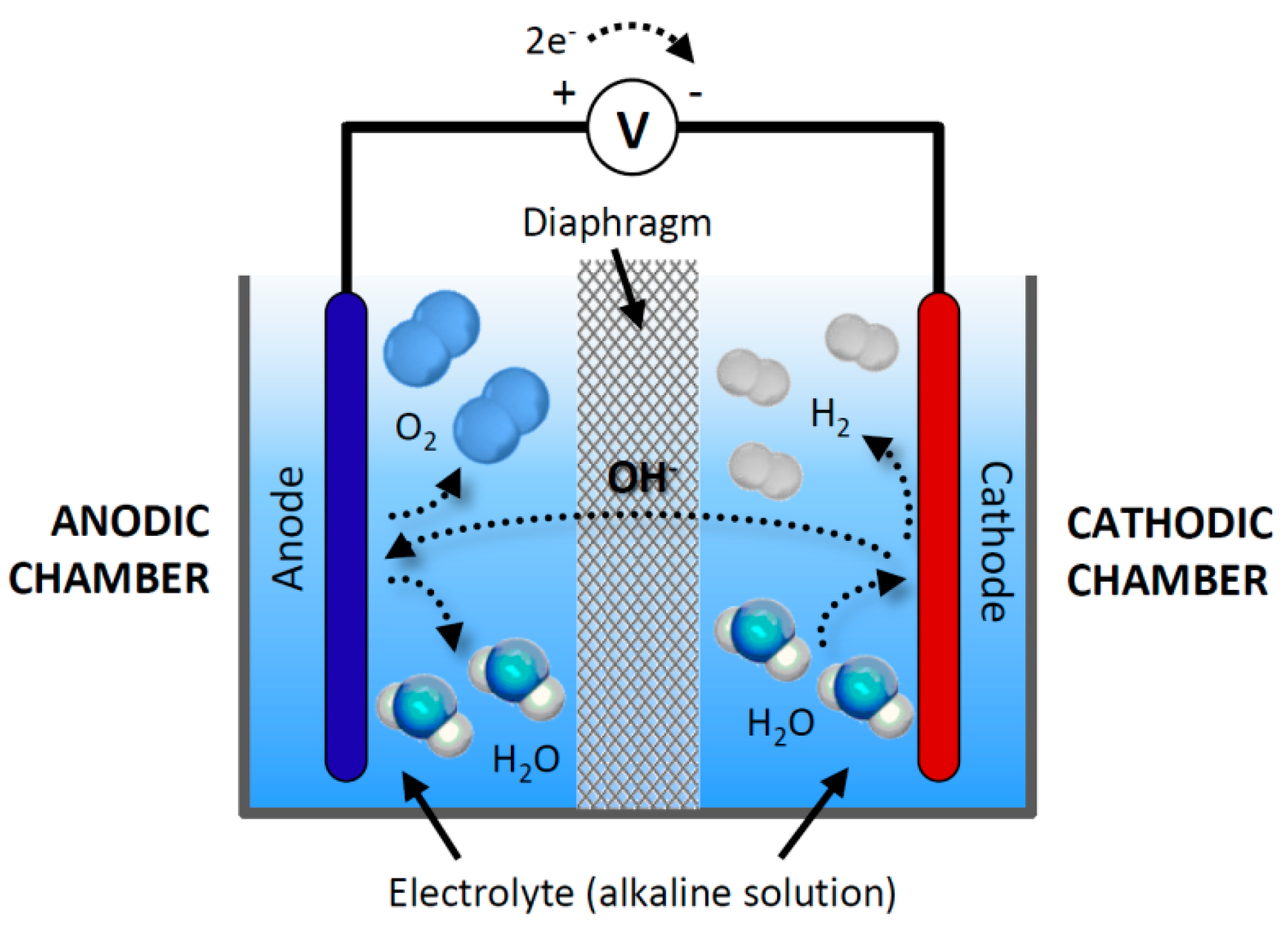
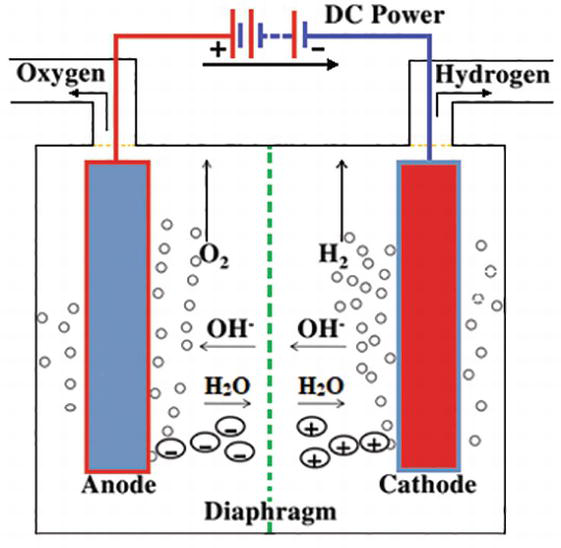
Water electrolysis is a quite old technology started around two centuries back, but promising technology for hydrogen production. ... The schematic diagram of alkaline water electrolysis is shown in Fig. 2. At the cathode side, molecules of water (H 2 O) were reduced to produce one...
15.1.1.1 Tests for Faraday's first law See diagram 3.3.4: Electrolysis Rinse a strip of thin copper cathode in deionized water. Dry and weigh to the nearest 0.01 g. Use a copper strip as anode. Add 200 g copper (II) sulfate crystals and 80 g concentrated sulfuric acid to 1 g of water.
Electrolysis of water. Electrolysing water splits the water molecules (H2O) into hydrogen (H2) and oxygen (O2) molecules according to the following From the diagram and equations, we can see that the volume of hydrogen produced is double the volume of oxygen. This confirms that there are twice...
August 22, 2018 - Hydrogen is a promising energy vector for the future. Among the different methods of its production, the electrolysis of water has attracted great attention because it is a sustainable and renewable chemical technology. Thus, hydrogen represents a suitable energy vector for the storage of ...
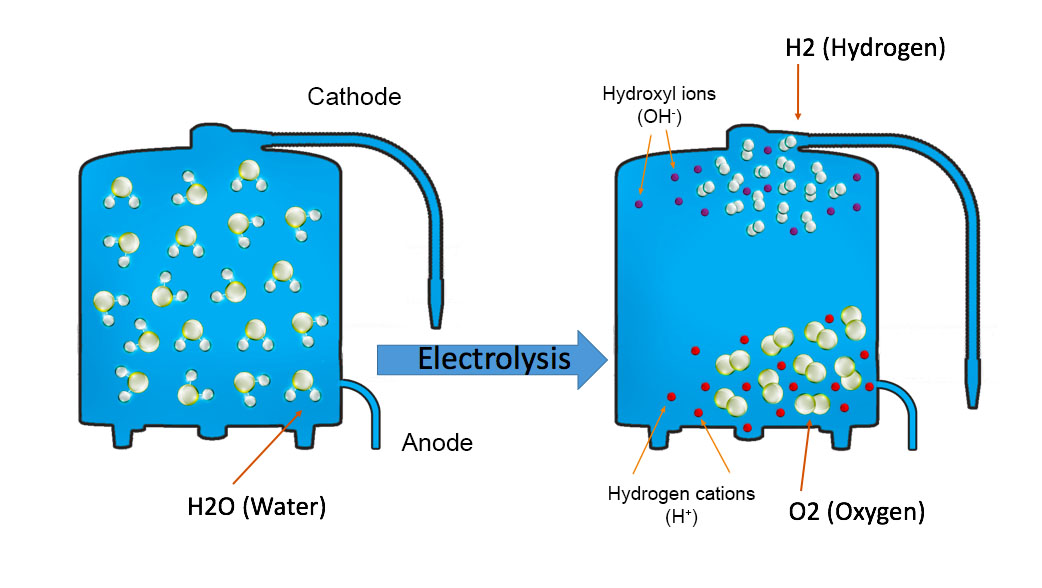
The chemical change that takes place in this activity is called the electrolysis of water, in which water gets decomposed into hydrogen and oxygen. First, water splits into hydrogen (H +) and hydroxide (OH –) ions. These lose charge to form hydrogen and oxygen at the electrodes. The overall reaction can be represented by
The electrolysis of water is a key component of any future energy systems aimed at achieving this goal. Hydrogen is an important industrial commodity. We are well on the way to achieving this goal. The diagram below shows the integration of electrolysis into a zero-emission future energy system.
March 29, 2020 -
If a container of water is subjected to electrolysis, hydroxyl and hydronium ions are produced which move through the liquid from one electrode to another. Rezwan Khan. Figure 3: Schematic diagram of the steps involved in treatment of floodwater by electrolysis. Following the above step another...
Download scientific diagram | Basic scheme of a water electrolysis system from publication: Hydrogen production by alkaline water electrolysis | Water electrolysis is one of the simplest methods used for hydrogen production. It has the advantage of being able to produce hydrogen using only ...
Water Electrolysis Process, Scientific Chemistry Diagram, Vector Illustration Educational Poster with power source,. Green Hydrogen, future energy ...
Open access peer-reviewed chapter. Electrolysis for Ozone Water Production. By Fumio Okada and Kazunari Naya. The formation of Pt oxides on the anode has been confirmed by the EPMA measurements. From the phase diagram of Pt in electrochemical equilibrium, PtO2 and PtO3 are...
The electrolysis of one mole of water produces a mole of hydrogen gas and a half-mole of oxygen gas in their normal diatomic forms. If solar or wind energy could be efficiently used to electrolyze water, the practicality of fuel cells would be enhanced.
Electrolysis of water is the process of using electricity to decompose water into oxygen and hydrogen gas by a process called electrolysis. Hydrogen gas released in this way can be used as hydrogen fuel, or remixed with the oxygen to create oxyhydrogen gas, which is used in welding and other applications. Sometimes called water splitting, electrolysis requires a minimum potential diffe…
April 8, 2020 - Water electrolysis is a reaction of the decomposition of a chemical molecule under the influence of an external electrical voltage ...
Background Electrolysis of water is the process by which water is decomposed into oxygen and hydrogen gas, when electric current is passed through it. Objectives To observe the electrolysis of water and determine the gases liberated at the end of experiment.
Hello Viewers, Welcome To BADARPUR DRAWING ACADEMY,Thank you for visiting, here this video will show you how to draw electrolysis of water diagram...
Illustration about Labeled diagram to show the electrolysis of acidified water forming hydrogen and oxygen gases. Illustration of diagram, acidified, electrode - 61461102
July 14, 2020 - With a sufficient potential difference, this may cause electrolysis with oxygen gas being produced at the anode and hydrogen gas produced at the cathode (see [1878] for current reviews). The electrolysis of water usually involves dilute, or moderately concentrated, salt solutions to reduce ...
28/01/2022 · The electrolysis of water is carried out in an electrolytic cell consisting of a pair of platinum electrodes immersed in water. To this water, a small amount of an electrolyte such as H 2 S O 4 has been added. The addition of an electrolyte is necessary because pure water lacks ions responsible for conducting electricity.























0 Response to "39 electrolysis of water diagram"
Post a Comment